STANDARDS:
HS-PS1-1: Use the periodic table as a model to predict the relative properties of elements based on the patterns of electrons in the outermost energy level atoms.
HS-PS1-2: Construct and revise an explanation for the outcome of a simple chemical reaction based on the outermost electron states of atoms, trends in the periodic table, and knowledge of the pattern of chemical properties.
HS-PS1-3: Plan and construct investigation to gather evidence to compare the structure of substance at the bulk scale to infer the strength of electrical forces between particles.
HS-PS1-7: Use mathematical representations to support the claim that atoms, and therefore mass, are conserved during a chemical reaction.
HS-PS3-1: Create a computational model to calculate the change of energy of one component in a system when the change in energy of the other component(s) and energy flows in a d out of the systems are known.
HS-PS3-2: Develop and use models to illustrate that energy at the macroscopic scale can be accounted for as a combination of energy associated with the motion of particles (objects) and energy associated with the relative position of particles (objects).
HS-PS3-4: Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperature are combined within a closed system results in a more uniform distribution among the components in the system (Second Law of Thermodynamics).
HS-PS3-5: Develop and use a model of two objects interacting through electric or magnetic fields to illustrate the force between objects and the changes in energy of the objects due to the interaction.
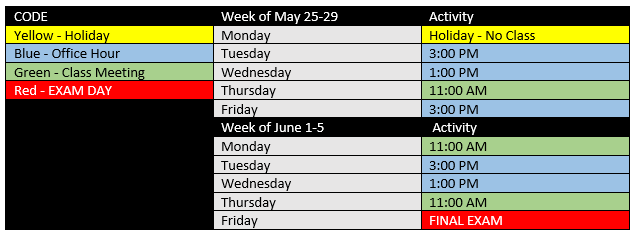
MEETING SCHEDULE
HS-PS1-1: Use the periodic table as a model to predict the relative properties of elements based on the patterns of electrons in the outermost energy level atoms.
HS-PS1-2: Construct and revise an explanation for the outcome of a simple chemical reaction based on the outermost electron states of atoms, trends in the periodic table, and knowledge of the pattern of chemical properties.
HS-PS1-3: Plan and construct investigation to gather evidence to compare the structure of substance at the bulk scale to infer the strength of electrical forces between particles.
HS-PS1-7: Use mathematical representations to support the claim that atoms, and therefore mass, are conserved during a chemical reaction.
HS-PS3-1: Create a computational model to calculate the change of energy of one component in a system when the change in energy of the other component(s) and energy flows in a d out of the systems are known.
HS-PS3-2: Develop and use models to illustrate that energy at the macroscopic scale can be accounted for as a combination of energy associated with the motion of particles (objects) and energy associated with the relative position of particles (objects).
HS-PS3-4: Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperature are combined within a closed system results in a more uniform distribution among the components in the system (Second Law of Thermodynamics).
HS-PS3-5: Develop and use a model of two objects interacting through electric or magnetic fields to illustrate the force between objects and the changes in energy of the objects due to the interaction.
MEETING SCHEDULE
ZOOM LINKS:
_ Regular class meeting: Tuesday and Thursday at 11 AM
https://lapromisefund.zoom.us/j/88298114478?pwd=YnhmbEF6L2RkNEtwT1RWUlJQemxxUT09
_ Review Office Hour:
Please check Google Classroom for more information.
TOPIC TO BE COVERED
NOTE: Calculate Reaction Rates is removed from the list, because we did not have time to cover it.
ASSIGNMENT:
Please complete the review packet and submit it to Google Classroom on Friday June 5th, by 9 PM.
_ Regular class meeting: Tuesday and Thursday at 11 AM
https://lapromisefund.zoom.us/j/88298114478?pwd=YnhmbEF6L2RkNEtwT1RWUlJQemxxUT09
_ Review Office Hour:
Please check Google Classroom for more information.
TOPIC TO BE COVERED
- Chemical foundation:
- Structures of an atom
- How to calculate particles within an isotopes
- How to write isotopes symbols
- Chemical bonding: know how to differentiate between different types of chemical bonding (covalent/molecular vs. ionic) using different methods.
- Composition (Metals or nonmetals)
- The difference in electronegativity
- How to read the periodic table: Know all the component of an elemental cell.
- Periodic table: understand the different between groups(columns) and period (rows)
- Periodic trends: (atomic radii, electron affinity, ionization energy, electronegativity) – Understand the definition of the terms.
- Nomenclature: Chemical naming
- Binary compound
- Compound with transition metal
- Acid
- Molecular/covalent compounds
- Write the chemical formula from given ions and from their names
- Percent Composition and Empirical Formula
- Lewis Dot Structure: know all the steps and how to apply them in drawing structures of given molecular compounds
- Molecular geometry: what are the shapes of compounds?
- Comparing bond angles.
- Know the name of all the shapes
- How to draw molecules in 3D using Lewis structure
- Polarity: is the compound polar or non-polar?
- Structures of an atom
- Chemical reaction
- Classification of chemical reaction
- Predict product(s) from given reactant(s)
- Balance chemical reaction
- Write complete chemical equation from given reactants’ names.
- Stoichiometry
- Understand the concept of mole
- Apply the Marvin’s Neighborhood too solve different quantities
- Convert grams to grams, mole to grams…
- Density
- Concentration (molarity)
- Know the unit of all quantities to avoid mixing them up
- How to calculate percent yield
- Know the difference between theoretical and experimental yield
- Thermodynamic:
- Energy transformation:
- Different forms of energy: initial and final form of energy in a system.
- Calorimetry
- Understand how to use the equation
- Know how to manipulate the equation to calculate different quantities
- Know how to apply the equation to calculate problems involving energy transfer (system in stoichiometry)
- Lattice heat of fusion and evaporation
- Bond Energy:
- Enthalpy vs. Entropy
- Gibbs Free Energy
- How to use the equation
- Manipulate the equation to calculate different quantities.
- Endothermic vs. Exothermic Process
- Equilibrium
- Use free energy to predict direction of chemical reaction (forward or reverse)
- Calculate reaction rates.
- Energy transformation:
- Other things to consider
- Vocabulary (mostly included in other sections)
- How to write CER
- Analyze lab data
- Lab technique and safety
NOTE: Calculate Reaction Rates is removed from the list, because we did not have time to cover it.
ASSIGNMENT:
Please complete the review packet and submit it to Google Classroom on Friday June 5th, by 9 PM.
| spring_final_review.docx | |
| File Size: | 33 kb |
| File Type: | docx |