STANDARDS:
HS-PS3-1: Create a computational model to calculate the change in the energy of one component in a system when the change in energy o the other component(s) and energy flows in and out of the system are known.
HS-PS3-2: Develop and use models to illustrate that energy at the macroscopic scale can be accounted for as a combination of energy associated with the motions of particles (objects) and energy associated with the relative position of particles (objects).
HS-PS3-4: Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperature are combined within a closed system results in a more uniform energy distribution among the components in the system.
OBJECTIVES:
_ Students will be able to calculate the total amount of heat substances need to change its temperature with the incorporation of energy required for phase change.
_ Students will be able to set up and calculate different quantities involved in a transfer of thermal energy between two objects within a closed system and make the connection to the Law of Conservation of Energy.
THREE DIMENSIONAL FOCUSES:
Science and Engineering Practice: Develop and use model based on evidence to illustrate the relationships between systems or between components of a system.
Disciplinary Core Ideas: Conservation of energy means that the total change o energy in any system is always equal to the total energy transferred into or out of the system
Crosscutting Concept: Energy cannot be created or destroyed – only moves between one place and another place, between objects and/or fields, or between systems.
PROCEDURES:
DO NOW ON GOOGLE CLASSROOM.
_ Log into your GOOGLE CLASSROOM and complete the "DO NOW: BASIC CALORIMETRY" assignment. (It will be available at 8 AM on Monday. If you login earlier, you won't be able to access the assignment)
IMPORTANCE: I will take attendance for Tuesday accordingly to your completion of this assignment.
PART 1: LATENT HEAT OF FUSION AND EVAPORATION
Definition:
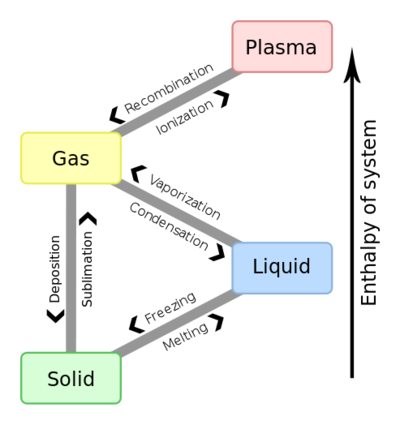
1. Latent heat: is the amount of heat absorbed/released by a substance during the process of changing from one phase (solid, liquid, or gas) to another.
2. Fusion: The process of changing from solid to liquid state of compounds.
3. Evaporation: The process of changing from liquid to gas state of compounds.
HS-PS3-1: Create a computational model to calculate the change in the energy of one component in a system when the change in energy o the other component(s) and energy flows in and out of the system are known.
HS-PS3-2: Develop and use models to illustrate that energy at the macroscopic scale can be accounted for as a combination of energy associated with the motions of particles (objects) and energy associated with the relative position of particles (objects).
HS-PS3-4: Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperature are combined within a closed system results in a more uniform energy distribution among the components in the system.
OBJECTIVES:
_ Students will be able to calculate the total amount of heat substances need to change its temperature with the incorporation of energy required for phase change.
_ Students will be able to set up and calculate different quantities involved in a transfer of thermal energy between two objects within a closed system and make the connection to the Law of Conservation of Energy.
THREE DIMENSIONAL FOCUSES:
Science and Engineering Practice: Develop and use model based on evidence to illustrate the relationships between systems or between components of a system.
Disciplinary Core Ideas: Conservation of energy means that the total change o energy in any system is always equal to the total energy transferred into or out of the system
Crosscutting Concept: Energy cannot be created or destroyed – only moves between one place and another place, between objects and/or fields, or between systems.
PROCEDURES:
DO NOW ON GOOGLE CLASSROOM.
_ Log into your GOOGLE CLASSROOM and complete the "DO NOW: BASIC CALORIMETRY" assignment. (It will be available at 8 AM on Monday. If you login earlier, you won't be able to access the assignment)
IMPORTANCE: I will take attendance for Tuesday accordingly to your completion of this assignment.
PART 1: LATENT HEAT OF FUSION AND EVAPORATION
Definition:
1. Latent heat: is the amount of heat absorbed/released by a substance during the process of changing from one phase (solid, liquid, or gas) to another.
2. Fusion: The process of changing from solid to liquid state of compounds.
3. Evaporation: The process of changing from liquid to gas state of compounds.
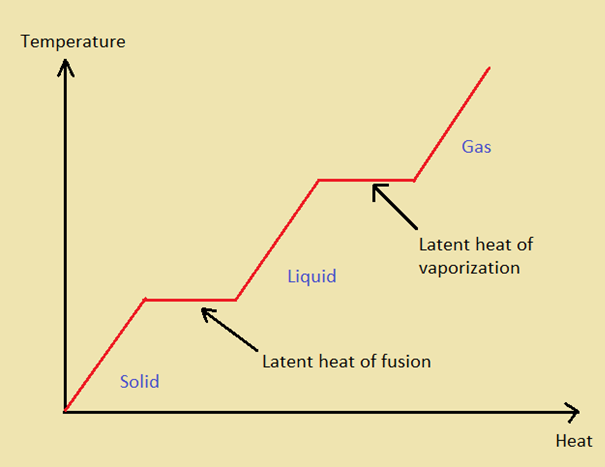
_ Your objective is to calculate the amount of heat a substance needs to reach certain temperature. The following diagram summarize this process
3. Lattice Energy of Fusion and Evaporation equation
q = n ΔHfus or q = n ΔHeva
In which
_ q is heat or energy (J)
_ n is mole of the compound (mol)
_ ΔHfus is heat of fusion (kJ/mol) and n ΔHeva is heat of evaporation (kJ/mol)
4. If you can't attend my online class, you can watch the following video to learn how the calculation is done:
https://www.khanacademy.org/science/chemistry/states-of-matter-and-intermolecular-forces/states-of-matter/v/specific-heat-heat-of-fusion-and-vaporization
5. If you attend my online class, we will work on the following examples:
Example 1: How much energy is required to melt 100 grams of water?
Example 2: How much energy is required to boil 150 grams of water?
Given that :
Phase Specific heat (J/gC) Phase Change Energy (kJ/mol)
Gas 2.02 Boiling 40.65
Liquid 4.184 Fusion 6.02
Solid 2.0
Example 3: How much energy is required to heat 200 grams of water from 25 oC to 125 oC?
Example 4: How much energy is required to heat 75 grams of water from -85 oC to 185 oC?
_ The following worksheet will be due on Tuesday at 9:00 PM on Google Classroom
q = n ΔHfus or q = n ΔHeva
In which
_ q is heat or energy (J)
_ n is mole of the compound (mol)
_ ΔHfus is heat of fusion (kJ/mol) and n ΔHeva is heat of evaporation (kJ/mol)
4. If you can't attend my online class, you can watch the following video to learn how the calculation is done:
https://www.khanacademy.org/science/chemistry/states-of-matter-and-intermolecular-forces/states-of-matter/v/specific-heat-heat-of-fusion-and-vaporization
5. If you attend my online class, we will work on the following examples:
Example 1: How much energy is required to melt 100 grams of water?
Example 2: How much energy is required to boil 150 grams of water?
Given that :
Phase Specific heat (J/gC) Phase Change Energy (kJ/mol)
Gas 2.02 Boiling 40.65
Liquid 4.184 Fusion 6.02
Solid 2.0
Example 3: How much energy is required to heat 200 grams of water from 25 oC to 125 oC?
Example 4: How much energy is required to heat 75 grams of water from -85 oC to 185 oC?
_ The following worksheet will be due on Tuesday at 9:00 PM on Google Classroom
| phase_change_energy_worksheet.docx | |
| File Size: | 13 kb |
| File Type: | docx |
_ Due on Wednesday: Please read the following document and take notes using my version of the Cornell's Note. Please make sure to include all the vocabularies and your summary must consist of at least 5 quality sentences.
_ Time: 11:59 PM
_ Time: 11:59 PM
| thermodynamics_text.pdf | |
| File Size: | 3897 kb |
| File Type: | |
| reading_cornells_note.docx | |
| File Size: | 14 kb |
| File Type: | docx |
PART 2: SYSTEM OF CALORIMETRY
IMPORTANT: After seeing what happened last week, I realized that the majority of you don't know how to perform one, two, or three step solving for variable. For that reason, please complete the following math practice. You will need this mathematical skill for this part of the lesson.
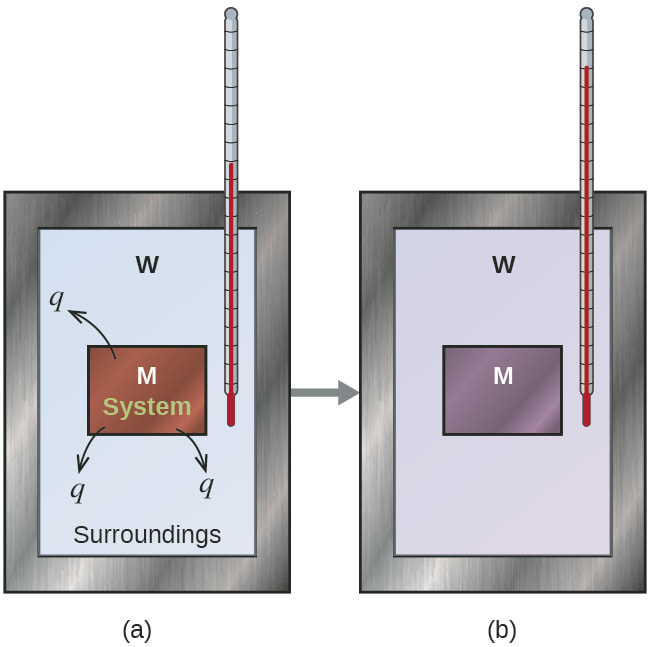
1. Observe the following image:
IMPORTANT: After seeing what happened last week, I realized that the majority of you don't know how to perform one, two, or three step solving for variable. For that reason, please complete the following math practice. You will need this mathematical skill for this part of the lesson.
- Login to IXL through Clever
- Under "Learning" tab, select "Algebra 1"
- Find "J - Solve Equation"
- Complete the following assignments
- #6 - Solving Equation With Variables on Both Sides
- #7 - Complete the Solution
1. Observe the following image:
In both images, a hot object (in red, labeled with M) is dropped into a tank of liquid or its surrounding (labeled with W). The thermometer is used to measure the temperature of the surrounding. In the second image, both object reach equilibrium (both have the same temperature)
_ The hot object cools down and transfers its heat to the surrounding
_ The surround heats up by absorbing the heat from the hot object.
If we write this in an equation:
The amount of heat released by the hot object = The amount of heat absorbed by the surrounding
or
- q_released = q_absorbed
_ For those who can't attend the zoom meeting, you can watch the following video for examples of how to solve the problems. (Stop at 37:38, you don't need the information beyond this point)
https://www.youtube.com/watch?v=ePm_N6RgLfk&t=590s
_ If you are up for the challenge, this is a really great reading about calorimetry. It's where I got the image from.
https://opentextbc.ca/chemistry/chapter/5-2-calorimetry/
_ Follow my lesson to solve the following examples:
Example 1: A 60 grams of solid sample at 100 degree Celsius is put into 150 g of water at room temperature (25 degree Celsius). Both the water the the solid sample reach equilibrium at 28 degree Celsius. What is the specific heat of the metal?
Example 2: A 3 grams sample of zinc is heated and placed in 70 grams of water. Temperature of water increases from 20 to 25 degree Celsius. Given that the specific heat of Zinc is 0.390 J/gC. What was the initial temperature of the zinc sample?
Example 3: A sample of silver with mass of 63 grams is heated to 110 degree Celsius and placed in a container of water at 20 degree Celsius. The final temperature of both objects is 23 degree Celsius. What is the mass of water in the container?
Example 4: If 40 grams of water at 70 degree Celsius is mixed with 40 gram ethanol at 10 degree Celsius, what is the final temperature of the mixture? Given that the specific heat of ethanol is 2.57 J/gC.
_ Complete the following worksheet and turn it into Google Classroom by 9:00 PM on Friday (April 24th)
OFFICE HOUR: I will have office hour at 3 PM on Friday. Please check Google Classroom for the zoom link.
_ The hot object cools down and transfers its heat to the surrounding
_ The surround heats up by absorbing the heat from the hot object.
If we write this in an equation:
The amount of heat released by the hot object = The amount of heat absorbed by the surrounding
or
- q_released = q_absorbed
_ For those who can't attend the zoom meeting, you can watch the following video for examples of how to solve the problems. (Stop at 37:38, you don't need the information beyond this point)
https://www.youtube.com/watch?v=ePm_N6RgLfk&t=590s
_ If you are up for the challenge, this is a really great reading about calorimetry. It's where I got the image from.
https://opentextbc.ca/chemistry/chapter/5-2-calorimetry/
_ Follow my lesson to solve the following examples:
Example 1: A 60 grams of solid sample at 100 degree Celsius is put into 150 g of water at room temperature (25 degree Celsius). Both the water the the solid sample reach equilibrium at 28 degree Celsius. What is the specific heat of the metal?
Example 2: A 3 grams sample of zinc is heated and placed in 70 grams of water. Temperature of water increases from 20 to 25 degree Celsius. Given that the specific heat of Zinc is 0.390 J/gC. What was the initial temperature of the zinc sample?
Example 3: A sample of silver with mass of 63 grams is heated to 110 degree Celsius and placed in a container of water at 20 degree Celsius. The final temperature of both objects is 23 degree Celsius. What is the mass of water in the container?
Example 4: If 40 grams of water at 70 degree Celsius is mixed with 40 gram ethanol at 10 degree Celsius, what is the final temperature of the mixture? Given that the specific heat of ethanol is 2.57 J/gC.
_ Complete the following worksheet and turn it into Google Classroom by 9:00 PM on Friday (April 24th)
OFFICE HOUR: I will have office hour at 3 PM on Friday. Please check Google Classroom for the zoom link.
| calorimetry_in_a_system.docx | |
| File Size: | 14 kb |
| File Type: | docx |
| calorimetry_in_a_system_spanish.docx | |
| File Size: | 14 kb |
| File Type: | docx |