STANDARDS:
HS-PS3-1: Create a computational model to calculate the change in the energy of one component in a system when the change in energy o the other component(s) and energy flows in and out of the system are known.
HS-PS3-2: Develop and use models to illustrate that energy at the macroscopic scale can be accounted for as a combination of energy associated with the motions of particles (objects) and energy associated with the relative position of particles (objects).
HS-PS3-4: Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperature are combined within a closed system results in a more uniform energy distribution among the components in the system.
OBJECTIVES:
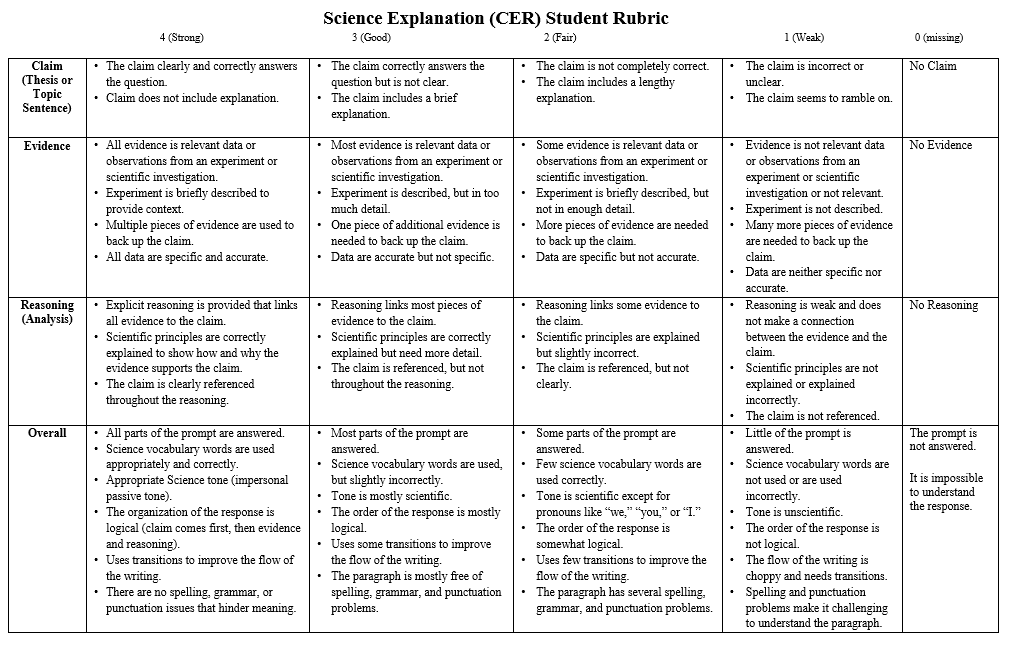
_ Students will be able to recognize common errors while writing CER and apply it to correct their own work.
_ Students will be able to calculate the bond enthalpy of given molecules.
_ Students will be able to calculate the change in enthalpy of given chemical reactions and determine whether the reaction is endothermic or exothermic.
THREE DIMENSIONAL FOCUSES:
Science and Engineering Practice:
_ Developing and Using Models: Develop and use a model based on evidence to illustrate the relationship between systems or between component of a system
Disciplinary Core Ideas:
_ PS3. B.2: Conservation of energy means that the total change of energy in any system is always equal to the total energy transferred into or out of the system.
_ PS3. B. 3: Energy cannot be created or destroyed, but it can be transported from one place to another and transferred between system.
Crosscutting Concept: Energy and Matter
_ Energy cannot be created or destroyed – only moves between one place and another place, between objects and/or fields, or between systems.
_ Systems and Systems Models: When investigating or describing a system, the boundaries and initial conditions of the system need to be defined and their inputs and outputs analyzed and described using models.
ZOOM LINK:
_ Monday, May 11th, 2020: https://lapromisefund.zoom.us/j/88298114478?pwd=YnhmbEF6L2RkNEtwT1RWUlJQemxxUT09
_ Thursday, May 14th, 2020: https://lapromisefund.zoom.us/j/88298114478?pwd=YnhmbEF6L2RkNEtwT1RWUlJQemxxUT09
PW: Chemistry
PROCEDURE:
PART 1: CER WORKSHOP
Step 1: Go over the following presentation.
https://docs.google.com/presentation/d/1KL85rUo0sTo8kUWy2sJT2zLQuQOn_CY9ZXliTqQFQkg/edit?usp=sharing
Step 2: Watch the recording video of the lecture recording (available after the class meeting).
Step 3: Work on correcting one of the CER. (You can work with a partner)
https://docs.google.com/document/d/1mZNOK6sC235LX5kJ0moxI57sE14r98Y8HoJlMw8WxYk/edit?usp=sharing
Step 4: Complete the CER assigned on Google Classroom. Make sure that you follow the rubric to avoid any unnecessary deduction.
HS-PS3-1: Create a computational model to calculate the change in the energy of one component in a system when the change in energy o the other component(s) and energy flows in and out of the system are known.
HS-PS3-2: Develop and use models to illustrate that energy at the macroscopic scale can be accounted for as a combination of energy associated with the motions of particles (objects) and energy associated with the relative position of particles (objects).
HS-PS3-4: Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperature are combined within a closed system results in a more uniform energy distribution among the components in the system.
OBJECTIVES:
_ Students will be able to recognize common errors while writing CER and apply it to correct their own work.
_ Students will be able to calculate the bond enthalpy of given molecules.
_ Students will be able to calculate the change in enthalpy of given chemical reactions and determine whether the reaction is endothermic or exothermic.
THREE DIMENSIONAL FOCUSES:
Science and Engineering Practice:
_ Developing and Using Models: Develop and use a model based on evidence to illustrate the relationship between systems or between component of a system
Disciplinary Core Ideas:
_ PS3. B.2: Conservation of energy means that the total change of energy in any system is always equal to the total energy transferred into or out of the system.
_ PS3. B. 3: Energy cannot be created or destroyed, but it can be transported from one place to another and transferred between system.
Crosscutting Concept: Energy and Matter
_ Energy cannot be created or destroyed – only moves between one place and another place, between objects and/or fields, or between systems.
_ Systems and Systems Models: When investigating or describing a system, the boundaries and initial conditions of the system need to be defined and their inputs and outputs analyzed and described using models.
ZOOM LINK:
_ Monday, May 11th, 2020: https://lapromisefund.zoom.us/j/88298114478?pwd=YnhmbEF6L2RkNEtwT1RWUlJQemxxUT09
_ Thursday, May 14th, 2020: https://lapromisefund.zoom.us/j/88298114478?pwd=YnhmbEF6L2RkNEtwT1RWUlJQemxxUT09
PW: Chemistry
PROCEDURE:
PART 1: CER WORKSHOP
Step 1: Go over the following presentation.
https://docs.google.com/presentation/d/1KL85rUo0sTo8kUWy2sJT2zLQuQOn_CY9ZXliTqQFQkg/edit?usp=sharing
Step 2: Watch the recording video of the lecture recording (available after the class meeting).
Step 3: Work on correcting one of the CER. (You can work with a partner)
https://docs.google.com/document/d/1mZNOK6sC235LX5kJ0moxI57sE14r98Y8HoJlMw8WxYk/edit?usp=sharing
Step 4: Complete the CER assigned on Google Classroom. Make sure that you follow the rubric to avoid any unnecessary deduction.
PART 2: ENTHALPY OF REACTION.
Step 1: Log into your www.explorelearning.com account, and click on the "Reaction Energy" activity (this is the same activity we did last week)
Step 2: You have two options (either one works)
1. Download a new document from the link below
2. Use the same document as you did last week.
Step 1: Log into your www.explorelearning.com account, and click on the "Reaction Energy" activity (this is the same activity we did last week)
Step 2: You have two options (either one works)
1. Download a new document from the link below
2. Use the same document as you did last week.
| reaction_energy.docx | |
| File Size: | 281 kb |
| File Type: | docx |
| reaction_energy_spanish.docx | |
| File Size: | 280 kb |
| File Type: | docx |
Step 3: Scroll down to activity C and set up the simulation according to the instruction
Step 4: Follow the instruction of Activity C to complete your assignment.
Step 5: Turn the assignment into Google Classroom by Friday May 15th at 9:00 PM.
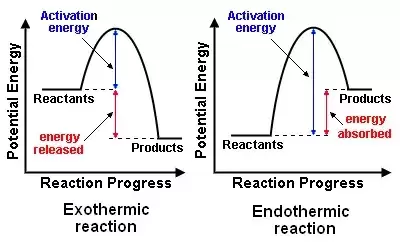
Step 6: Observe the following graphs
Step 4: Follow the instruction of Activity C to complete your assignment.
Step 5: Turn the assignment into Google Classroom by Friday May 15th at 9:00 PM.
Step 6: Observe the following graphs
Vocabulary:
_ Reactants/Products: Input and output of a chemical reaction.
_ Activation energy: energy required to start a reaction. If the reaction doesn't meet this energy level, it won't occur.
_ Energy released: energy lost to the environment (exothermic process), feel hot to the touch, and the enthalpy of reaction is negative.
_ Energy absorbed: energy taken in by the reaction (endothermic process), feel cold to the touch, and enthalpy of reaction is positive.
Step 6: Watch the following video
https://www.youtube.com/watch?v=xE1gdQZcR-o
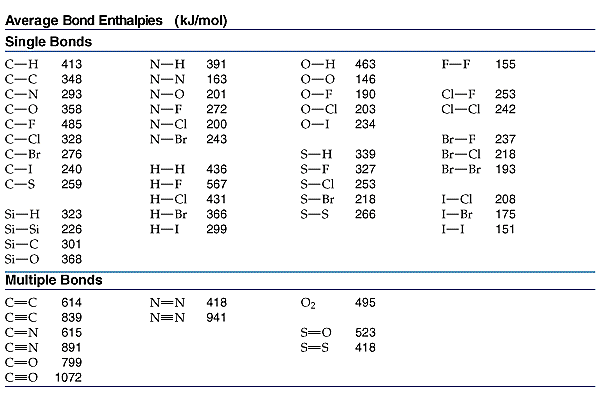
_ Use the following data as your references.
_ Reactants/Products: Input and output of a chemical reaction.
_ Activation energy: energy required to start a reaction. If the reaction doesn't meet this energy level, it won't occur.
_ Energy released: energy lost to the environment (exothermic process), feel hot to the touch, and the enthalpy of reaction is negative.
_ Energy absorbed: energy taken in by the reaction (endothermic process), feel cold to the touch, and enthalpy of reaction is positive.
Step 6: Watch the following video
https://www.youtube.com/watch?v=xE1gdQZcR-o
_ Use the following data as your references.
Step 7: Watch my instructional video from Google Classroom if you did not watch the video in step 6.
_ Example 1: Calculate the enthalpy of reaction for the chemical reaction between hydrogen and fluorine gas to form Hydrogen Fluoride.
_ Example 2: Calculate the enthalpy of reaction for the combustion of Ethanol (C2H5OH).
Step 8: Complete the following worksheet and submit to Google Classroom by 9:00 PM on Friday.
_ Example 1: Calculate the enthalpy of reaction for the chemical reaction between hydrogen and fluorine gas to form Hydrogen Fluoride.
_ Example 2: Calculate the enthalpy of reaction for the combustion of Ethanol (C2H5OH).
Step 8: Complete the following worksheet and submit to Google Classroom by 9:00 PM on Friday.
| bond_energy_ws.docx | |
| File Size: | 50 kb |
| File Type: | docx |