STANDARDS:
HS-PS3-1: Create a computational model to calculate the change in the energy of one component in a system when the change in energy o the other component(s) and energy flows in and out of the system are known.
HS-PS3-2: Develop and use models to illustrate that energy at the macroscopic scale can be accounted for as a combination of energy associated with the motions of particles (objects) and energy associated with the relative position of particles (objects).
HS-PS3-4: Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperature are combined within a closed system results in a more uniform energy distribution among the components in the system.
OBJECTIVES:
_ Students will be able to understand the understand the concept of entropy and other components in the Gibbs free energy calculation.
_ Students will be able to manipulate and calculate different quantities in the equation.
THREE DIMENSIONAL FOCUSES:
Science and Engineering Practice:
_ Developing and Using Models: Develop and use a model based on evidence to illustrate the relationship between systems or between component of a system
Disciplinary Core Ideas:
_ PS3. B.2: Conservation of energy means that the total change of energy in any system is always equal to the total energy transferred into or out of the system.
_ PS3. B. 3: Energy cannot be created or destroyed, but it can be transported from one place to another and transferred between system.
Crosscutting Concept: Energy and Matter
_ Energy cannot be created or destroyed – only moves between one place and another place, between objects and/or fields, or between systems.
_ Systems and Systems Models: When investigating or describing a system, the boundaries and initial conditions of the system need to be defined and their inputs and outputs analyzed and described using models.
ZOOM LINK:
_ Monday, May 11th, 2020: https://lapromisefund.zoom.us/j/88298114478?pwd=YnhmbEF6L2RkNEtwT1RWUlJQemxxUT09
_ Thursday, May 14th, 2020: https://lapromisefund.zoom.us/j/88298114478?pwd=YnhmbEF6L2RkNEtwT1RWUlJQemxxUT09
PW: Chemistry
PROCEDURE:
PART 1: INTRODUCTION TO FREE ENERGY
Step 1: Watch the following video:
https://ed.ted.com/lessons/what-is-entropy-jeff-phillips
+ As you watch the video, focus on finding the answer for the following questions:
1. What is entropy?
2. How does entropy react in a system?
Step 2: You have two options for this one.
_ Option 1: Watch my lecture video in case you don't show up to class.
_ Option 2: Watch the following video to help guide you through how to use the Gibbs free energy equation
https://www.khanacademy.org/science/chemistry/thermodynamics-chemistry/gibbs-free-energy/v/gibbs-free-energy-and-spontaneity
Note: I simplified the equation down while planning my lesson. This video provides more details about each quantity in the equation.
Gibbs' Free Energy: ΔG = ΔH - TΔS
1. ΔH: The change in Enthalpy. We learned how to do this in our previous lesson. Unit is kJ.
2. ΔS: Change in Entropy. This value are likely to be given to you. Unit is kJ/K
3. T: Temperature in Kelvin.
K = C + 273.15
4. ΔG: Free energy. Unit is kJ
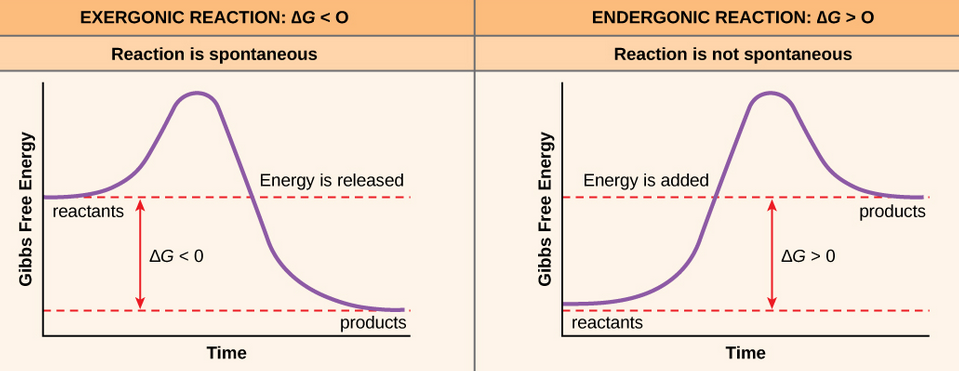
_ This value dictates whether a chemical reaction will be forward or reverse.
+ ΔG < 0: The reaction is spontaneous or moving forward
+ ΔG > 0: The reaction is unspontaneous or reverse
+ ΔG = 0: The reaction is at equilibrium (We will learn about this next week)
HS-PS3-1: Create a computational model to calculate the change in the energy of one component in a system when the change in energy o the other component(s) and energy flows in and out of the system are known.
HS-PS3-2: Develop and use models to illustrate that energy at the macroscopic scale can be accounted for as a combination of energy associated with the motions of particles (objects) and energy associated with the relative position of particles (objects).
HS-PS3-4: Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperature are combined within a closed system results in a more uniform energy distribution among the components in the system.
OBJECTIVES:
_ Students will be able to understand the understand the concept of entropy and other components in the Gibbs free energy calculation.
_ Students will be able to manipulate and calculate different quantities in the equation.
THREE DIMENSIONAL FOCUSES:
Science and Engineering Practice:
_ Developing and Using Models: Develop and use a model based on evidence to illustrate the relationship between systems or between component of a system
Disciplinary Core Ideas:
_ PS3. B.2: Conservation of energy means that the total change of energy in any system is always equal to the total energy transferred into or out of the system.
_ PS3. B. 3: Energy cannot be created or destroyed, but it can be transported from one place to another and transferred between system.
Crosscutting Concept: Energy and Matter
_ Energy cannot be created or destroyed – only moves between one place and another place, between objects and/or fields, or between systems.
_ Systems and Systems Models: When investigating or describing a system, the boundaries and initial conditions of the system need to be defined and their inputs and outputs analyzed and described using models.
ZOOM LINK:
_ Monday, May 11th, 2020: https://lapromisefund.zoom.us/j/88298114478?pwd=YnhmbEF6L2RkNEtwT1RWUlJQemxxUT09
_ Thursday, May 14th, 2020: https://lapromisefund.zoom.us/j/88298114478?pwd=YnhmbEF6L2RkNEtwT1RWUlJQemxxUT09
PW: Chemistry
PROCEDURE:
PART 1: INTRODUCTION TO FREE ENERGY
Step 1: Watch the following video:
https://ed.ted.com/lessons/what-is-entropy-jeff-phillips
+ As you watch the video, focus on finding the answer for the following questions:
1. What is entropy?
2. How does entropy react in a system?
Step 2: You have two options for this one.
_ Option 1: Watch my lecture video in case you don't show up to class.
_ Option 2: Watch the following video to help guide you through how to use the Gibbs free energy equation
https://www.khanacademy.org/science/chemistry/thermodynamics-chemistry/gibbs-free-energy/v/gibbs-free-energy-and-spontaneity
Note: I simplified the equation down while planning my lesson. This video provides more details about each quantity in the equation.
Gibbs' Free Energy: ΔG = ΔH - TΔS
1. ΔH: The change in Enthalpy. We learned how to do this in our previous lesson. Unit is kJ.
2. ΔS: Change in Entropy. This value are likely to be given to you. Unit is kJ/K
3. T: Temperature in Kelvin.
K = C + 273.15
4. ΔG: Free energy. Unit is kJ
_ This value dictates whether a chemical reaction will be forward or reverse.
+ ΔG < 0: The reaction is spontaneous or moving forward
+ ΔG > 0: The reaction is unspontaneous or reverse
+ ΔG = 0: The reaction is at equilibrium (We will learn about this next week)
Step 3: Watch another video - This one help simplify the understanding about entropy for you
https://www.khanacademy.org/science/biology/energy-and-enzymes/the-laws-of-thermodynamics/v/introduction-to-entropy
Step 4: Consider the following faction: (Refer the questions to the equation)
1. How does increasing/decreasing enthalpy affect the sign of free energy?
2. How does increasing/decreasing temperature affect the sign of free energy?
3. How does increasing/decreasing entropy affect the sign of free energy?
Step 5: Work on the worksheet and submit to Google Classroom by 9:00 PM on Tuesday (05/19)
https://www.khanacademy.org/science/biology/energy-and-enzymes/the-laws-of-thermodynamics/v/introduction-to-entropy
Step 4: Consider the following faction: (Refer the questions to the equation)
1. How does increasing/decreasing enthalpy affect the sign of free energy?
2. How does increasing/decreasing temperature affect the sign of free energy?
3. How does increasing/decreasing entropy affect the sign of free energy?
Step 5: Work on the worksheet and submit to Google Classroom by 9:00 PM on Tuesday (05/19)
| gibbs_free_energy_ws.docx | |
| File Size: | 17 kb |
| File Type: | docx |
PART 2: MANIPULATION OF QUANTITIES IN FREE ENERGY CALCULATION
Step 1: Please watch my instructional video of how you can manipulate and calculate different quantities in the free energy equation
Step 2: Complete the worksheet and submit to Google Classroom by 9:00 PM on Friday (05/22)
Step 1: Please watch my instructional video of how you can manipulate and calculate different quantities in the free energy equation
Step 2: Complete the worksheet and submit to Google Classroom by 9:00 PM on Friday (05/22)
| gibbs_free_energy_2.docx | |
| File Size: | 16 kb |
| File Type: | docx |